Response to health alert
Project leader: Marc Grandadam
Background
In March 2014, the Ebola virus emerged in four Western African countries, leading to an unprecedented situation as the virus spread from selvatic foci to urban areas. Difficulties in containing the epidemics led the World Health Organization (WHO) to implement surveillance and alert programs worldwide to prevent a pandemic expansion of the virus. The Institut Pasteur du Laos has been mandated by the Lao Ministry of Health to set up a laboratory plan to investigate locally suspected cases. The transfer of viral hemorrhagic fever virus diagnostic tools has been performed with the help of the French WHO collaborative centre for arbovirus and viral hemorrhagic fevers co-hosted by the Institut Pasteur teams in Paris and Lyon. A response team has been trained to handle samples under high security conditions on a 24/7 basis (Figure n°2). Internal and external global alert exercises have been organized to test lab capacities and coordination between the partners in the surveillance system. This capacity building has been partially supported by the WHO Western Pacific office. The laboratory response capacity has been assessed by organizing full-size internal and external exercises. The mean time to obtain a first RT-PCR result is 5 hours 30 minutes.
Consulting activities
IP Laos teams were asked to carry out a risk assessment of French schools in Vientiane (Lycée Hoffet and the French nursery school) to provide recommendations for vector control methods. On-site visits were organized in the presence of the different school staff including management, the bursar, housekeepers and gardeners. Alternative strategies were proposed to reduce the use of chemical compounds in order to minimize the toxic exposure of staff and children and the emergence of insecticide resistance in mosquitoes.
Support activities
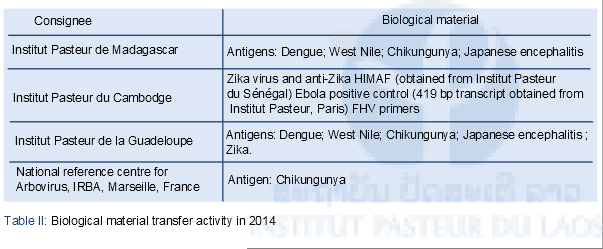
The A&EVD laboratory benefited from the support of members of the International Pasteur Network (IP Sénégal, IP Madagascar, IP Hong Kong and NIHE, Hanoi) to launch its research activities. As technical platforms are now fully operational, the lab can now respond to requests for reagents and reference materials from other Institutes within the Pasteur Network. Table II summarizes biological material transfers in 2014.