SATREPS Project: Project for Development of Innovative Research Techniques in the Genetic Epidemiology of Malaria and Other Parasitic Diseases in the Lao PDR for Containment of Their Expanding Endemicity
Project coordinator: Dr. Shigeyuki Kano
Member of staff: Moritoshi Iwagami, Phonepadith Xangsayarath, Phonepadith Khattignavong, Sengdeuane Keomalaphet, Pheovaly Soundala, Lavy Lorphachan, and other visiting scientists and students
Background:
Malaria, Schistosomiasis (Schistosoma mekongi), and Opisthorchiasis (Opisthorchis viverrini) are a tremendous health burden on the people of the Lao PDR. Although significant reductions in malaria transmission have been reported due to the large-scale distribution of insecticide-treated bed nets (ITNs) through the Global Fund to Fight AIDS, Tuberculosis and Malaria, strategies based on the scientific evidence have not been developed to deal with the genetic variations in parasite and vector populations, and drug-resistant malaria. Recently, artemisinin-resistant malaria was reported in southern provinces (Ashley et al., 2014, Ménard et al., 2016). Therefore, it is necessary to survey other provinces, especially in the southern part of the country, to monitor and contain the spread of drug-resistant malaria. The Lao Ministry of Health and the WHO set a goal of eliminating malaria by 2030. To achieve this goal, we have to understand the real malaria situation, including that of drug-resistant malaria, and develop elimination strategies.
Since Schistosomiasis (S. mekongi) and Opisthorchiasis (O. viverrini) are localized to the Lao PDR and surrounding countries, they are recognized as neglected tropical diseases. Nevertheless, the prevalence of Opisthorchiasis is estimated to be as high as 15–54% in the Lao PDR. Little information on the molecular/genetic epidemiology of Opisthorchiasis is available to develop effective measures for prevention and diagnosis of the disease.
The Government of the Lao PDR requested the Japan International Cooperation Agency (JICA) to establish the Lao–Japan Joint Laboratory within Institut Pasteur du Laos (IPL) to conduct highly technological research on human malaria parasites: Plasmodium falciparum, P. vivax; the monkey malaria parasite: P. knowlesi; and human trematodiasis: S. mekongi and O. viverrini. The joint research will concentrate on genetic epidemiological studies to detect and control the emergence and dissemination of these parasitic diseases. The project also contributes to the capacity development of researchers and technicians in the Lao PDR through training in field and lab work, seminars, and career development.
In order to carry out this project, the IPL collaborates with the National Center for Global Health and Medicine (NCGM), Tokyo, Japan, the Center of Malariology, Parasitology and Entomology (CMPE), the National Institute of Public Health (NIOPH), and other Departments of the Ministry of Health, Lao PDR.

Objective:
The objectives of this project are (1) to develop more convenient and accurate methods (PCR methods, LAMP methods, etc.) for diagnosis of the diseases, (2) to monitor the temporal and spatial epidemiological situations of pathogens and vectors of the diseases, (3) to analyze mechanisms of emergence and expansion of drug-resistant malaria, especially artemisinin resistance, and (4) to analyze the glucose-6-phosphate dehydrogenase (G6PD) activity of the Lao population for evaluation of the possible usage of primaquine ( Howes et al., 2013), utilizing molecular biological techniques. Based on the scientific evidence obtained by this project, health education for the people will be strengthened and the endemicity of the diseases will be monitored together with the local Lao Ministry of Health. Research results will also be utilized in government services for the sustainable development of the Lao PDR.
Study period of the project:
Five years (May 2014 to April 2019)
Study sites of the project:
Malaria:
Savannakhet province, Saravane province, Sekong province, Attapeu province, Champasak province, Khammouane province, Phongsali province, Luang Prabang province.
Schistosomiasis (S. mekongi): Khong district and Mounlapamok district, Champasak province.
Opisthorchiasis (O. viverrini): Khammouane province, Champasak province.
Ethical clearance:
The SATREPS project was approved by the National Ethics Committee for Health Research in the National Institute of Public Health (NIOPH), Ministry of Health, Lao PDR in 2014, 2015, 2016 (extended each year).
Activities and Results in November 2015 to October 2016:
We conducted seven field surveys on parasitic diseases, and six refresher training courses in parasitic disease diagnoses for medical lab technicians in the five southern provinces (Savannakhet, Saravane, Sekong, Champasak and Attapeu) since November 2015. We also implemented a malaria DNA diagnosis system (LAMP method) at Central level (CMPE) and three sentinel provinces (Luang Prabang, Savannakhet and Champasak) in 2016. This activities report summarizes our activities and the results of the SATREPS project in the Lao–Japan Parasitology lab from November 2015 to October 2016.
All field surveys were conducted in collaboration with the CMPE, Provincial Health Offices, and District Health Offices, Ministry of Health, Lao PDR. All the training courses were also conducted in collaboration with the CMPE and Provincial Health Offices.
Field surveys on Malaria, Schistosomiasis, and Opisthorchiasis:
1. Malaria Survey for blood sample collection in five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu), 18th October to 1st November 2015.
2. Malaria and G6PD activity survey, Khong district, Mounlapamok district, Phatounphone district, Champasak province, 1st to 13th February 2016.
3. Malaria Survey for blood sample collection in five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu), 17th to 25th April 2016.
4. Schistosomiasis and Opisthorchiasis Survey, Khong district, Champasak province, 24th to 30th April 2016.
5. Opisthorchiasis Survey, Yommalath district, Khammouane province, 16th to 28th June 2016.
6. Schistosomiasis Survey, Khong district and Mounlapamok district, Champasak province, 4th September to 17th October 2016.
7. Malaria Survey for blood sample collection in five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu), 29th October to 5th November, and 14th November 2016.
Field surveys:
Written informed consent was obtained from all the participants prior to interviews and biological sample collection. The guardians of child participants consented to their participation. Three malaria diagnostic methods— microscopy, a rapid diagnostic test (RDT: Malaria Ag Pf / Pv, Standard Diagnostics, Inc. Republic of Korea), and a DNA diagnostic method—were used for detecting malaria parasites, malaria parasites antigen, and malaria parasites DNA, respectively. The Kato-Katz thick smear method of stool examination was used for detecting the eggs of S. mekongi and O. viverrini in the stool samples.
1. Malaria Survey for blood sample collection in the five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu), 18th October to 1st November 2015
We collected 4,439 dried blood samples on filter papers (FTATM Classic Card, GE Healthcare Life Sciences, WhatmanTM, UK) from malaria suspected patients who came to 69 public health facilities on the five southern provinces. The 69 public health facilities were provincial hospitals, district hospitals, and health centers in malaria high endemic districts selected based on a report on malaria cases in 2013 summarized by the CMPE. A summary of the samples is shown in Table 1. DNA was extracted from all the samples using a QIAamp DNA Mini Kit (QIAGEN, Co. Japan). The DNA is now being analyzed by PCR* at IPL to detect malaria parasites and identify the species. After finishing the species identification, we will analyze drug-resistant mutation(s) in the K13 gene (artemisinin-resistant gene) and pfcrt gene (chloroquine-resistant gene) of Plasmodium falciparum, also at IPL.
*PCR: Polymerase Chain Reaction
Table 1. Summary of blood samples collected in the five southern provinces in 2015.
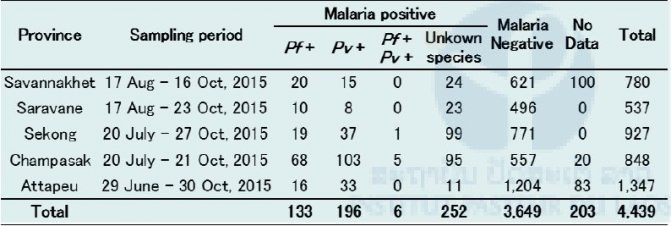
Pf: Plasmodium falciparum, Pv: P. vivax
We analyzed the patients’ data in each province and found that the malaria positivity rate of males was significantly higher than that of females in all provinces (P < 0.01) except Savannakhet (Figures 1–10). Moreover, the 15–39-year-old male population tended to be malaria positive, and the occupation of most was agriculture. These results suggest that the malaria high-risk population in the four provinces was the adult male population, and this seemed to be associated with their working style, i.e. they routinely work on their farms, as well as in forests for collecting food and wood. The current malaria elimination program is basically a household-based approach. Our result suggests that a job-based approach is also needed for malaria elimination in the Lao PDR.
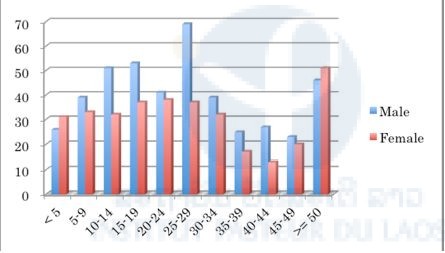
Figure 1. Number of malaria suspected cases in selected public health facilities in Savannakhet Province (17th August–16th October 2015).
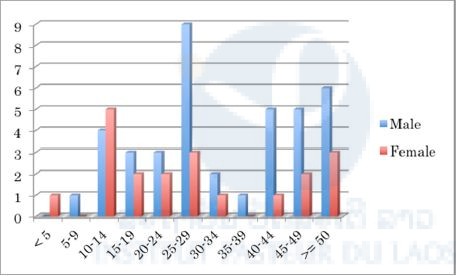
Figure 2. Number of malaria positive cases in selected public health facilities in Savannakhet Province (17th August–16th October 2015).
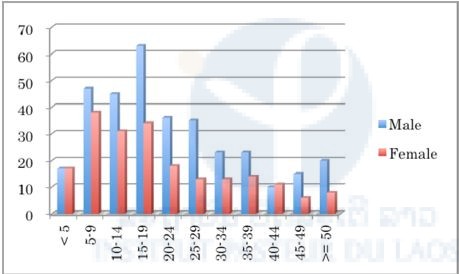
Figure 3. Number of malaria suspected cases in selected public health facilities in Saravane Province (17th August–23rd October 2015).
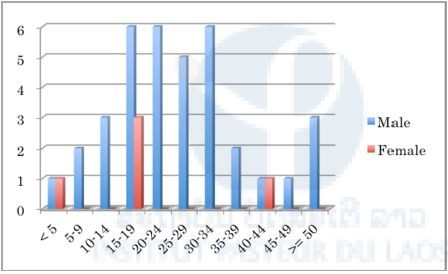
Figure 4. Number of malaria positive cases in selected public health facilities in Saravane Province (17th August–23rd October 2015).
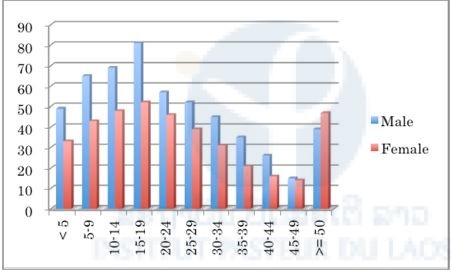
Figure 5. Number of malaria suspected cases in selected public health facilities in Sekong Province (20th July–27th October 2015).
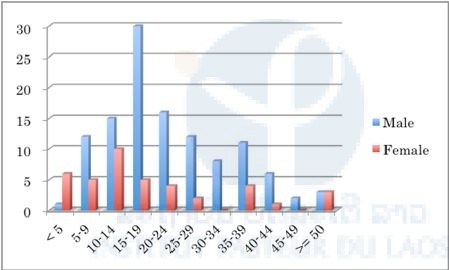
Figure 6. Number of malaria positive cases in selected public health facilities in Sekong Province (20th July–27th October 2015).
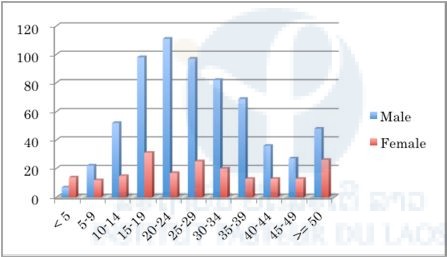
Figure 7. Number of malaria suspected cases in selected public health facilities in Champasak Province (20th July–21st October 2015).
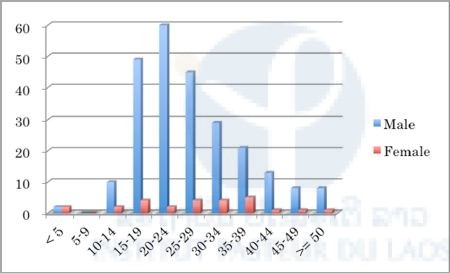
Figure 8. Number of malaria positive cases in selected public health facilities in Champasak Province (20th July-21st October 2015).
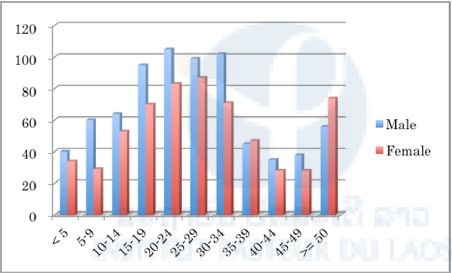
Figure 9. Number of malaria suspected cases in selected public health facilities in Attapeu Province (29th June–30th October 2015).
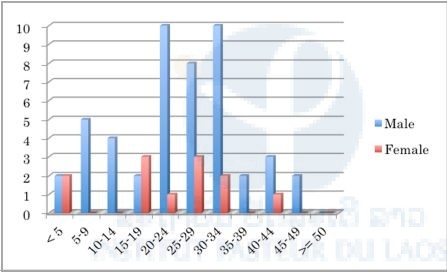
Figure 10. Number of malaria positive cases in selected public health facilities in Attapeu Province (29th June–30th October 2015).
2. Malaria and G6PD* Activity Survey, Khong district, Mounlapamok district, Phatounphone district, Champasak province, 1st–13th February 2016.
*G6PD: Glucose-6-phosphate dehydrogenase
Six hundred ninety-four villagers participated in this survey. Blood samples were collected by finger prick using a lancet. Malaria infection was checked by malaria rapid diagnostic test (RDT; Malaria Ag P.f/P.v, Standard Diagnostics, Inc., Republic of Korea) on site. When the villagers were diagnosed with malaria, a staff member from a district hospital or health center provided them with one pack of an antimalarial drug (Coartem). Blood smears and dried blood samples on filter papers were also obtained from the villagers. They were analyzed by a microscope and PCR, respectively, at IPL. A summary of the malaria survey by malaria RDT and PCR are shown in Tables 2 and 3, respectively.
Table 2. Summary of malaria survey and results of malaria RDT in Champasak province in February 2016.
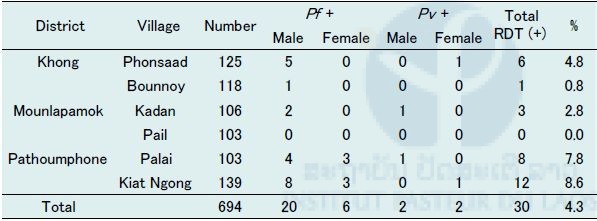
Pf: Plasmodium falciparum, Pv: P. vivax
Table 3. Summary of malaria survey and results of malaria PCR in Champasak province in February 2016.
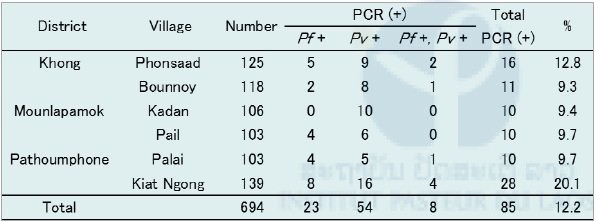
Pf: Plasmodium falciparum, Pv : P. vivax
G6PD enzyme activity was measured on site by a G6PD assay kit (Dojindo Molecular Technologies, Inc., Japan) and a spectrophotometer (NanoDrop 2000c, Thermo Fisher Scientific, Inc., USA). Reagents for G6PD assay were stored in a portable liquid nitrogen tank (Vapor Shipper SC 4/3v, MVE Co., USA). When G6PD enzyme activity was less then 60% compared to a positive control (G6PD enzyme activity normal blood), mutation(s) of the G6PD gene was examined by PCR and DNA sequencing by a Genetic Analyzer 3500XL (Applied Biosystems brand, Thermo Fisher Scientific, Inc., USA) at IPL. We failed to measure G6PD activity in 4 blood samples out of 694 blood samples due to technical problems. A summary of G6PD survey is shown in Table 4 and Figures 11 and 12.
Table 4. Participants in the malaria G6PD survey in Champasak province in February 2016.
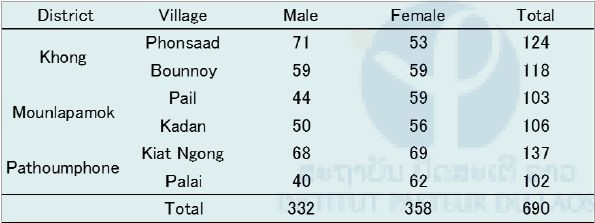
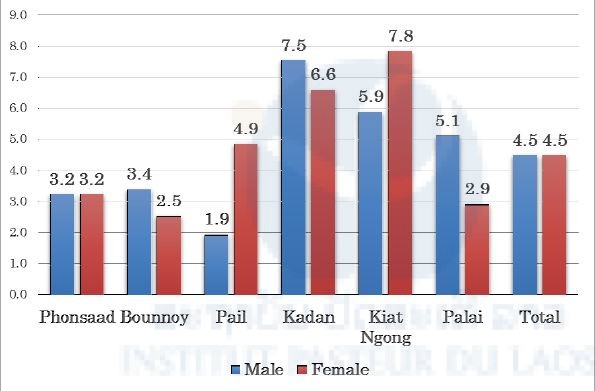
Figure 11. Percentages of G6PD deficiency among participants in this survey in Champasak province in February 2016.
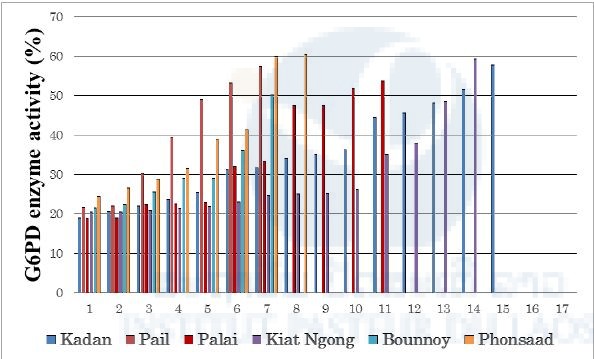
Figure 12. Percentages of G6PD enzyme activities among G6PD-deficient participants in this survey in Champasak province in February 2016.
G6PD enzyme activity between 10%–60% compared to normal G6PD enzyme activity is considered as moderate deficiency (WHO 1989). Therefore, all G6PD-deficient people in this survey were classified as moderately deficient. In this study, 61 people were diagnosed with G6PD deficiency and 4 people’s blood samples could not be measured for G6PD enzyme activity due to technical problems. Then, DNA was extracted from the blood samples from these 65 people and examined for mutation(s) of the G6PD gene by PCR and DNA sequencing at IPL. PCR primer sets from a previous study were used (Matsuoka et al. 2007). The results showed that the Viangchan (Vientiane)-type mutation in exon 9 of the G6PD gene (Matsuoka et al. 2004) was observed in 25 samples (3.6%). A heterogeneous genotype, i.e. a mixture of the Vientiane-type mutation and the wild type (normal), was also observed in 17 samples (2.5%). The female has two X-chromosomes and their G6PD genotype is sometimes heterogeneous because the G6PD gene is encoded in the X-chromosome in human beings. Sixteen of the seventeen samples were female, one was male.
3. Malaria Survey for blood sample collection in the five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu), 17th to 25th April 2016.
We collected 2,409 dried blood samples on filter papers (FTATM Classic Card, GE Healthcare Life Sciences, WhatmanTM, UK) from malaria patients who were diagnosed by microscopy or malaria RDT at 156 public health facilities on the five southern provinces. A summary of the samples is shown in Table 5. All the blood samples were sent to the Department of Tropical Medicine and Malaria (Dr. Kano’s laboratory; Dr. Kano is a chief advisor to the SATREPS project), Research Institute, National Center for Global Health and Medicine (NCGM), Tokyo, Japan on 27th July 2016.
Malaria DNA analysis is now being conducted at the NCGM lab using those filter paper samples. First, malaria parasite species have been identified by a nested real-time PCR. Second, mutations of drug-resistant associated genes, such as the K13 gene, or pfcrt gene, are being examined using P. falciparum-positive samples. Third, population genetic analyses will be conducted using highly polymorphic DNA markers of malaria parasites, such as microsatellite DNA loci, to understand the transmission dynamics of malaria in the Lao PDR.
An authorization for sending blood samples from the Lao PDR to Japan was obtained by Dr. Bouasy Hongvanthong, CMPE director and SATREPS project director. A Memorandum of Understanding (MOU) for collaborative research was signed between NCGM and IPL before starting the SATREPS project. A Material Transfer Agreement (MTA) was included in the MOU. The samples were sent to Japan
Table 5. Summary of malaria blood samples collected in April 2016 in five southern provinces.
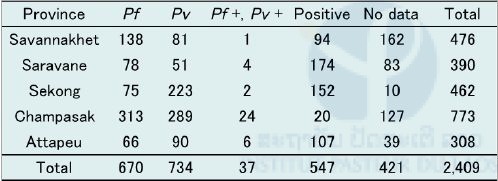
Pf: Plasmodium falciparum, Pv : P. vivax
The K13 gene (artemisinin-resistant associated gene) of P. falciparum is now being analyzed at NCGM, Tokyo. In Saravane province, 86 out of 142 P. falciparum isolates (62.7%) possessed C580Y mutation in the K13 gene while in Savannakhet province, 2 out of 58 P. falciparum isolates (3.4%) possessed C580Y mutation in the gene. C580Y means an amino acid substitution from cytosine (C) to tyrosine (Y) at codon 580 on the K13 gene. This preliminary data suggests heterogeneity of distribution patterns in artemisinin-resistance in the Lao PDR. This data was reported to the Lao Ministry of Health in November 2016.
4. Schistosomiasis and Opisthorchiasis Survey, Khong district, Champasak province, 24th to 30th April 2016
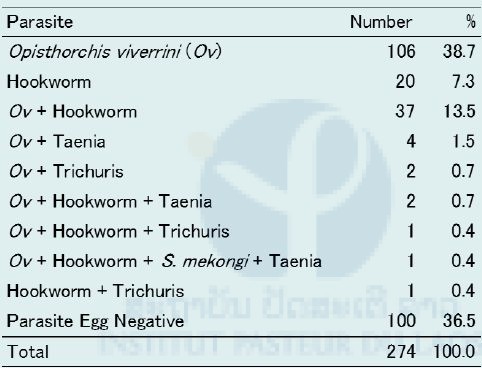
We collected 274 stool samples from 3 villages (Khon Neua, Khon Tai, and Hang Khon) in Khon Island and also collected 3,115 intermediate snail hosts, Neotricula aperta, at 6 riversides in Khon Neua village in the island (Figure 13). Stool samples were examined by the Kato-Katz thick smear method (three slides/sample) at IPL (Table 6). One hundred fifty-three stool samples were O. viverrini-egg positive (56.5%) while only one stool sample was S. mekongi-egg positive (0.4%) by the Kato-Katz method. The S. mekongi patient was infected with O. viverrini, Hookworm, and Taenia.
Table 6. Summary of Kato–Katz Stool examination on Khon Island, Khong district, Champasak province in April 2016.
Aliquots of all stool samples and all the snail samples were preserved in 70% ethanol and sent to Dr. Kumagai’s laboratory (Dr. Kumagai is a member of the SATREPS project), Tokyo Medical and Dental University (TMDU), Tokyo, Japan on 5th July 2016. Dr. Kumagai developed the S. mekongi LAMP method. The stool samples were analyzed by the LAMP method for detecting S. mekongi DNA at the TMDU lab and eight samles were found positive (Table 7).
Table 7. Result of S. mekongi LAMP examination using stool samples from Khon Island, Khong district, Champasak province in April 2016.
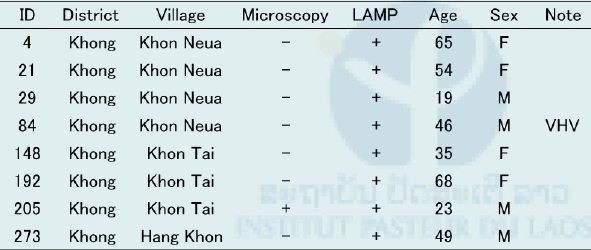
VHV: Village Health Volunteer
DNA was extracted from the pooled snail samples with a maximum of 200 per pool by the alkali-boiling method. The extracted DNA was directly applied to the LAMP method for S. mekongi, which detected positive snails infected with S. mekongi at a prevalence rate of over 0.5% in Khon Island. A prevalence rate over 0.5% means more than 1 positive snail out of 200 snails per pool. The geographical locations of snail collection sites (six sites) and the houses of six villagers who were positive by the LAMP method were plotted on Google Maps (Figure 13). The location of the houses of two villagers who were positive by the LAMP method could not be identified in this survey.
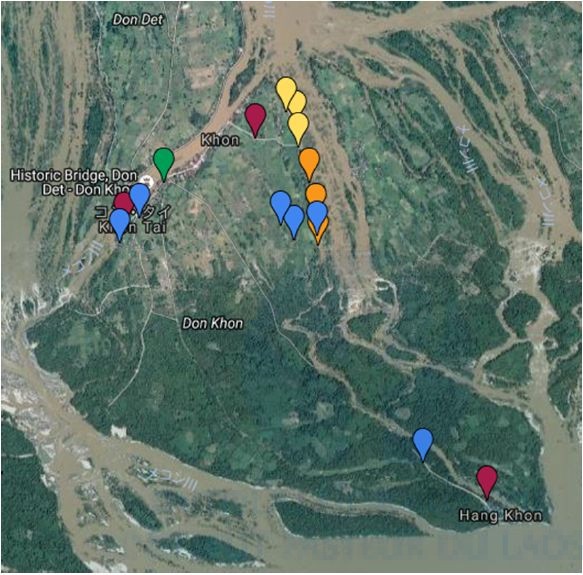
Figure 13. Geographical location of Ban Khon Health Center
(green), three study sites (villages, wine red), collection sites of S. mekongi negative snails (yellow), collection sites of S. mekongi positive snails (orange), houses of S. mekongi patients (LAMP +, blue), Google Maps Co.
Authorization for sending stool and snail samples from the Lao PDR to Japan was obtained by Dr. Bouasy Hongvanthong, CMPE director and project director of the SATREPS project. An MTA was obtained between TMDU and IPL before the samples were sent to Japan by a shipping company (FedEx) with appropriate packing.
5. Opisthorchiasis Survey, Yommalath district, Khammouane province, 16th to 28th June 2016
In the previous study in Yommalath district in 2015, we found that the surfaces of cutting boards in the house of an Opisthorchis viverrini-negative family was clean whereas the surfaces of cutting boards in the houses of O. viverrini-positive families were dirty. There were many fish scales on the surface, i.e. the O. viverrini-positive family didn’t wash their cutting board after use. In order to monitor the contamination situation of O. viverrini metacercariae on the surfaces of cutting boards in the endemic areas, we conducted a cutting-board study on O. viverrini with a molecular diagnosis technique (LAMP method). To standardize the condition of the cutting boards, we distributed new cutting boards to 62 households (two cutting boards for each household) in Yommalath district two weeks before this survey, and asked the villagers to use the new cutting boards for their cooking. The target households were selected based on previous studies in 2015, including both O. viverrini-positive and O. viverrini-negative families.
- Figure 14. Scratching a cutting board with a cooking knife and clean drinking water.
- Figure 15. Filtration of the water containing the sediments from the cutting boards.
We collected sediments from the surfaces of the cutting boards by scratching them with a cooking knife in the villagers’ kitchens and using clean drinking water (Figure 14). Then, the water containing the sediments from the surfaces of the cutting boards was filtered on site using a nylon mesh (43µm diameter) and a vacuum pump (Figure 15 and 16). The pore size of the nylon mesh is smaller than that of O. viverrini metacercariae. The nylon mesh containing the sediments was kept in 70% ethanol until DNA extraction. DNA was extracted from the nylon mesh by the alkali-boiling method at IPL. The extracted DNA was directly applied to the LAMP method for O. viverrini. No O. viverrini DNA was detected from the surfaces of cutting boards in this study.

Figure 16. Filtration of the water containing the sediments from the cutting boards with a vacuum pump.
6. Schistosomiasis Survey, Khong district and Mounlapamok district, Champasak province on 4th September to 17th October 2016
To eliminate Schistosomiasis mekongi in the Lao PDR, WHO, the Lao Ministry of Health, IPL, and other partners made a strategic plan for its elimination as a public health problem in 2016. To monitor the prevalence of schistosomiasis in the Lao PDR, seven sentinel villages and seven spot-check villages were selected based on previous surveys conducted by the Lao Ministry of Health. The seven sentinel villages will not change every year while the seven spot-check villages will be randomly selected every year from among 202 endemic villages in Champasak province.
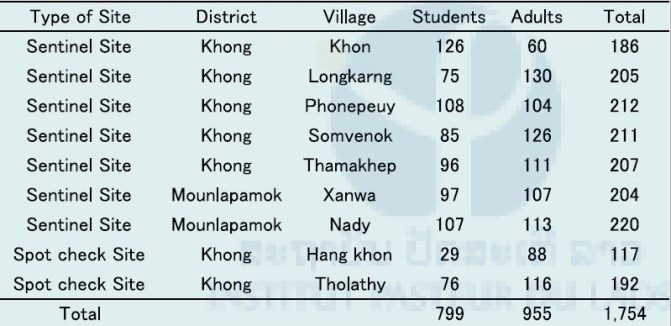
The Lao Ministry of Health and IPL conducted a collaborative survey in the seven sentinel villages (Table 8) and two of seven spot-check villages (Table 9) in 2016. Stool samples and blood samples were collected from villagers. The stool samples were examined on site by the Kato-Katz thick smear method (2 slides/sample) by Lao Ministry of Health staff, and the blood samples will be examined by the ELISA technique at IPL to detect S. mekongi antibodies using S. japonicum egg antigen. A total of 1,851 stool samples were collected from the seven sentinel villages and two spot-check villages and 43 samples were positive for S. mekongi eggs (Table 8 and 9). Therefore, the average prevalence of S. mekongi was 2.3% (43/1,851) in this survey. A total of 1,754 blood samples were collected from villagers in the seven sentinel villages and two spot-check villages (Table 10).
On 18th October, Dr. Kumagai from TMDU, Japan, who is a member of the SATREPS project, conducted Schistosoma ELISA training for IPL staff using schistosomiasis-positive and schistosomiasis-negative blood samples (IPL staff).
Table 8. Summary of S. mekongi survey in seven sentinel villages in Champasak province during September to October 2016.
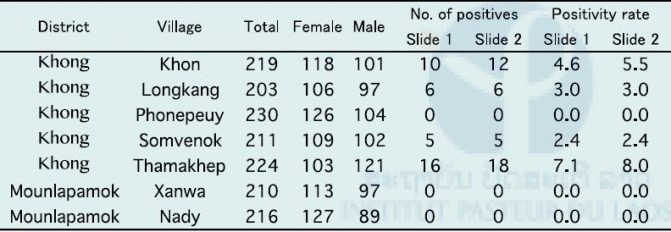
Table 9. Summary of S. mekongi survey in two spot-check villages in Champasak province in October 2016.
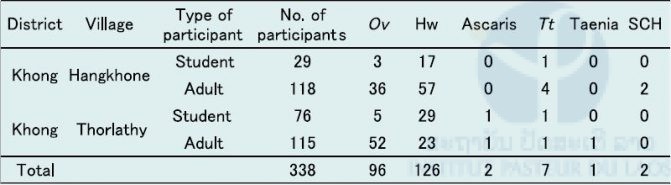
Ov: O. viverrini, Hw: hookworm, Tt: Trichuris trichiura, SHC: S. mekongi
Table 10. Number of blood samples collected in seven sentinel villages and two spot-check villages for schistosomiasis in Champasak province during September to Octobler 2016.
7. Malaria Survey for blood sample collection in the five
southern provinces (Savannakhet, Saravane, Sekong, Champasak and Attapeu), 29th October to 5th November, and 14th November 2016
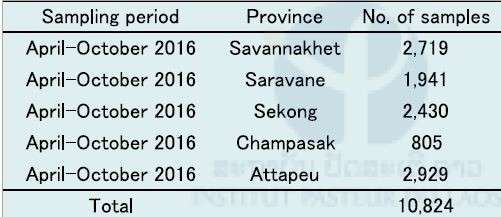
We collected 10,824 dried blood samples on filter papers (FTATM Classic Card, GE Healthcare Life Sciences, WhatmanTM, UK) from malaria suspected patients who came to public health facilities on the five southern provinces in October and November 2016. The sampling period was April to October 2016. The number of blood samples in each province is shown in Table 11.
Table 11. Number of malaria suspected blood samples collected in the five southern provinces in October and November, 2016.
Field training:
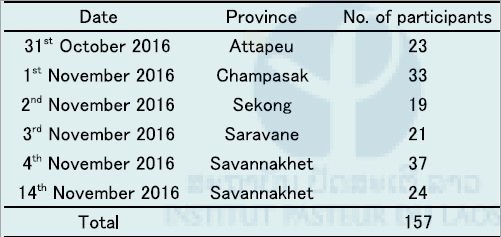
Six refresher training courses on parasitic disease diagnoses for medical lab technicians in the five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu) were conducted when we collected malaria suspected blood samples in the five provinces in October and November 2016. One refresher training course per province was conducted, except for Savannakhet province where two training courses were conducted because there were too many participants (61 people). A total of 157 medical lab technicians in public health facilities participated in the refresher training courses (Table 12).
Table 12. Summary of six refresher training courses in the five southern provinces in 2016.
In the training, we reviewed the contents of a previous training course in 2015 and updated the current malaria situation in the Lao PDR including the drug-resistant malaria situation in the Lao PDR and Greater Mekong Sub-region. We also reported on results of the analyses of malaria blood samples previously collected by the participants in October 2015 and April 2016. We plan to conduct refresher courses for the medical lab technicians in the five southern provinces once a year during the period of the SATREPS project.
Implementation of malaria DNA diagnosis system:
Previously, our malaria studies demonstrated that there were several asymptomatic carriers (parasite reservoirs or hidden malaria) in the endemic areas. Moreover, most of those cases cannot be detected by standard diagnosis techniques, i.e. microscopy and RDT. As mentioned above, the Lao Ministry of Health and WHO have set a goal to eliminate malaria by 2030 in the Lao PDR. To achieve this goal, some highly sensitive malaria diagnosis techniques are needed in this country.
In fact, one section, “Strategy 2.6 Strengthening the quality assurance and control systems for malaria diagnosis and antimalarial drugs” on page 33 of the “National Strategic Plan for Malaria Control and Elimination 2016-2020”, says “CMPE will also work with technical partners to develop molecular diagnostic capacity at selected provincial laboratories to ensure capacity for more sensitive diagnostic testing.” According to this plan, IPL and the SATREPS project decided to implement a malaria molecular diagnostic technique (LAMP method) in laboratories at central and provincial level.
At central level, a laboratory in the CMPE was selected for the malaria LAMP system, while at provincial level, the Station of Malariology, Parasitology and Entomology (SMPE), in Provincial Health Offices in Luang Prabang, Savannakhet and Champasak, were selected. These three provinces are sentinel provinces for the monitoring and control of malaria in the country.
- CMPE, Vientiane Capital, 15th–17th August 2016
- SMPE, Savannakhet Provincial Office, 22nd–23rd
- SMPE, Champasak Provincial Office, 24th–25th August 2016
- SMPE, Luang Prabang Provincial Office, 5th–7th September 2016
The Malaria LAMP system (LoopampTM MALARIA Pan/Pf Detection Kit, LoopampTM PURE DNA Extraction Kit and LoopampTM LF-160 heat block with UV detector), developed by the Eiken Chemical Co., Japan, was employed for this purpose because this system is highly sensitive to “nested malaria PCR,” is easy to operate, requires only one machine, and all reagents can be kept at room temperature. First, IPL staff translated the LAMP system Standard Operation Procedure (SOP) from English to Lao. Then, IPL staff trained three CMPE laboratory staff members at IPL in August 2016. This training course was composed of lectures and practical work. At the end of the training course, all the equipment (LoopampTM LF-160, a micro-pipet), reagents (LoopampTM Malaria Pan/Pf Detection Kit (48 tests X 3), LoopampTM PURE DNA Extraction Kit (90 tests X 2), 334mM NaCl solution, a UV-protective goggles) and some consumables (filtered pipet chips, tissue papers, filter papers for blood collection, plastic bags with zip locks, desiccants, a hand-punch) were installed at the laboratory in the CMPE (Figure 17).

Figure 17. Implementation of Malaria LAMP system at laboratory in CMPE in August 2016.
IPL staff and CMPE staff conducted implementation of the LAMP system in the three provinces in August and September 2016. Four laboratory staff in SMPE Savannakhet, two in SMPE Champasak, and one in Champasak Provincial Hospital participated in the training course. Four laboratory staff in SMPE Luang Prabang and one in Luang Prabang Provincial Hospital also participated in the training course, which was composed of lectures and practical work. The LAMP systems are now being utilized daily to diagnose malaria suspected patients, especially in the cases where microscopy and malaria RDT are negative but a patient has malaria-like symptoms and signs, such as recurring fever and shivering.
We plan to conduct a “Refresher course in Malaria LAMP Techniques” for laboratory staff in the CMPE and three SMPEs in the three provinces at least once a year during the period of SATREPS project.
Summary:
We conducted seven field surveys on parasitic diseases, six refresher training courses in parasitic disease diagnoses for medical lab technicians in the five southern provinces (Savannakhet, Saravane, Sekong, Champasak, and Attapeu) and implemented the malaria DNA diagnosis system (LAMP method) at Central level (CMPE) and in three sentinel provinces (Luang Prabang, Savannakhet, and Champasak) in 2016. This activities report summarizes our activities and the results of the SATREPS project in the Lao–Japan Parasitology Lab from November 2015 to October 2016.
Malaria study:
We collected malaria suspected blood samples from 156 public health facilities in the five southern provinces on October–November 2015 (4,439 samples), April 2016 (2,409 samples) and October–November 2016 (10,824 samples). We analyzed patient data for each of these provinces in October–November 2015 and found that the male malaria positivity rate was higher than that of females except in Savannakhet province. Moreover, the adult male population tended to be malaria positive. This result suggests that the malaria high-risk population is the adult male population in those four southern provinces.
Eighty-six out of 142 P. falciparum isolates (62.7%) collected in Saravane province on April 2016 possessed a mutation (C580Y) in the K13 gene that is responsible for artemisinin resistance. On the other hand, 2 out of 58 P. falciparum isolates (3.4%) collected in Savannakhet province in April 2016 possessed the mutation (C580Y) in the K13 gene. This result demonstrates heterogeneity in the distribution patterns of artemisinin resistance in the Lao PDR. Careful monitoring of the efficacy of artemisinin is necessary, especially in Saravane province.
The G6PD survey in Champasak province in February 2016 demonstrated that 8.8% of villagers (61/690) in the malaria-endemic areas were G6PD-deficient and their G6PD enzyme activities were between 20%–60% (moderate deficiency) compared to G6PD-normal blood. Twenty-five people (3.6%) have the “Viangchan type mutation” in the G6PD gene and 17 people (2.5%) have mixed genotypes with the “Viangchan type mutation” and “normal” in the G6PD gene.
Schistosomiasis study:
We developed the Schistosoma mekongi LAMP method and compared the sensitivity of the LAMP method and the Kato-Katz thick smear method. We examined 274 stool samples collected from villagers in Khon Island, Khong district, Champasak province in April 2016 by the two methods. By the Kato-Katz thick smear method (3 slides/sample), only one positive case was detected, while by the LAMP method, eight positive cases were detected from the 274 stool samples. Therefore, the sensitivity of the LAMP method is eight times higher than that of the Kato-Katz method. We also examined 3,115 freshwater snails (Neotricula aperta), an intermediate host of S. mekongi, collected from six riversides on Khon Island in April 2016, by the LAMP method. This estimated that the positivity rate of the snail samples was 0.5% at three riversides and 0.0% at the other three.
The Lao Ministry of Health and IPL conducted a collaborative schistosomiasis survey in September–October 2016. Stool examination by the Kato-Katz thick smear method (2 slides/sample) was conducted on site at seven sentinel villages and two spot-check villages. A total of 1,851 stool samples were examined and 43 samples were positive for S. mekongi eggs by the Kato-Katz thick smear method. Therefore, the prevalence of S. mekongi was 2.3% in this survey.
Opisthorochiasis study:
We attempted to detect O. viverrini metacercariae DNA on the surfaces of cutting boards in houses in Yommalath district, Khammouane province in June 2016. We selected 62 households, including both O. viverrini-positive and O. viverrini-negative families. To standardize cutting board conditions, we distributed two new cutting boards to each household two weeks before the survey and asked villagers to use them for daily cooking. Cutting board surfaces were scratched with a cooking knife and clean drinking water applied. This water was filtered by nylon mesh with a pore size (43µm diameter) smaller than the size of O. viverrini metacercariae. DNA was extracted from the sediments from the nylon mesh and the O. viverrini LAMP method was applied. No O. viverrini DNA was detected from the cutting board surfaces in this study.
Refresher training courses:
In 2015, we conducted training courses for parasitic disease diagnoses for medical lab technicians in 157 public health facilities in the five southern provinces at IPL and in the five southern provinces. We conducted six refresher training courses for the participants when we collected malaria suspected blood samples in the five provinces in October and November 2016.
Implementation of malaria LAMP system:
The Malaria LAMP System (DNA diagnosis method) was implemented at a laboratory in the CMPE and three laboratories in the Stations of Malariology, Parasitology and Entomology (SMPEs) in the Provincial Health Offices in Luang Prabang, Savannakhet, and Champasak provinces in August and September 2016. IPL staff conducted training courses in the malaria LAMP method. The malaria LAMP method is now utilized as a routine diagnosis method in the laboratories. Refresher training courses will be conducted once a year.
Partners:
- Center of Malariology, Parasitology and Entomology (CMPE), Ministry of Health, Vientiane Capital, Lao PDR
- National Institute of Public Health (NIOPH), Ministry of Health, Vientiane Capital, Lao PDR
- Department of Communicable Diseases Control (DCDC), Ministry of Health, Vientiane Capital, Lao PDR
- Department of Training and Research, Ministry of Health, (DTR) Vientiane Capital, Lao PDR
- Department of Hygiene and Health Promotion (DHHP), Ministry of Health, Vientiane Capital, Lao PDR
- National Center for Global Health and Medicine (NCGM), Tokyo, Japan
- Department of Community and Global Health, School of International Health, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan
- Department of Global Health, Graduate School of Health Sciences, University of the Ryukyus, Okinawa, Japan
- Department of Molecular and Cellular Parasitology, Juntendo University School of Medicine, Tokyo, Japan
- Section of Environmental Parasitology, Department of International Health Development, Division of Public Health, Graduate School, Tokyo Medical and Dental University, Tokyo, Japan.
Scientific communications:
Publications:
Ménard D, Khim N, Beghain J, Adegnika AA, Shafiul-Alam M, Amodu O, Rahim-Awab G, Barnadas C, Berry A, Boum Y, Bustos MD, Cao J, Chen JH, Collet L, Cui L, Thakur GD, Dieye A, Djallé D, Dorkenoo MA, Eboumbou-Moukoko CE, Espino FE, Fandeur T, Ferreira-da-Cruz MF, Fola AA, Fuehrer HP, Hassan AM, Herrera S, Hongvanthong B, Houzé S, Ibrahim ML, Jahirul-Karim M, Jiang L, Kano S, Ali-Khan W, Khanthavong M, Kremsner PG, Lacerda M, Leang R, Leelawong M, Li M, Lin K, Mazarati JB, Ménard S, Morlais I, Muhindo-Mavoko H, Musset L, Na-Bangchang K, Nambozi M, Niaré K, Noedl H, Ouédraogo JB, Pillai DR, Pradines B, Quang-Phuc B, Ramharter M, Randrianarivelojosia M, Sattabongkot J, Sheikh-Omar A, Silué KD, Sirima SB, Sutherland C, Syafruddin D, Tahar R, Tang LH, Touré OA, Tshibangu-wa-Tshibangu P, Vigan-Womas I, Warsame M, Wini L, Zakeri S, Kim S, Eam R, Berne L, Khean C, Chy S, Ken M, Loch K, Canier L, Duru V, Legrand E, Barale JC, Stokes B, Straimer J, Witkowski B, Fidock DA, Rogier C, Ringwald P, Ariey F, Mercereau-Puijalon O; KARMA Consortium. A Worldwide Map of Plasmodium falciparum Artemisinin Resistance. New England Journal of Medicine. 374:2453–2464,2016
Pongvongsa T, Nonaka D, Iwagami M, Nakatsu M, Phongmany P, Nishimoto F, Kobayashi J, Hongvanthon B, Brey PT, Moji K, Mita T, Kano S. Household clustering of asymptomatic malaria infections in Xepon district, Savannakhet province, Lao PDR. Malaria Journal. 15:508, 2016.
Oral presentations:
Lorphachan L, Soundala P, Keomalaphet S, Khattingavong P, Sengphimthong S, Iwagami M, Hongvanthong B, Brey PT, Kano S. Training course in Malaria and Parasitic Diseases Diagnosis at five southern provinces in the Lao PDR. The 10th National Health Research Forum, Savannakhet province, Lao PDR, October 27th-28th, 2016.
Keomalaphet S, Khattignavong P, Soundala P, Lorphachan L, Xangsayalath P, Iwagami M, Hongvanthong B, Brey PT, Kano S. Malaria Epidemiology in five southern provinces: Savannakhet, Saravane, Sekong, Champasak and Attapeu in the Lao PDR in 2015. The 10th National Health Research Forum, Savannakhet province, Lao PDR, October 27th-28th, 2016.
Ong KIC, Iwagami M, Lorphachane L, Soundala P, Xangsayarath P, Hongvanthong B, Brey PT, Kano S, Jimba M. Glucose-6-phosphate dehydrogenase (G6PD) deficiency among people in malaria endemic areas in Champasak Province, Lao PDR. The 10th National Health Research Forum, Savannakhet province, Lao PDR, October 27th–28th, 2016.
Araki H, Ong KIC, Lorphachane L, Soundala P, Iwagami M, Hongvanthong B, Brey PT, Kano S, Jimba M. Factors associated with Opisthorchis viverrini infection among children in an endemic area in Khammnouane province, Lao PDR. The 10th National Health Research Forum, Savannakhet province, Lao PDR, October 27th–28th, 2016.
Kumagai T, Iwagami M, Yamabe M, Keomalaphet S, Khattignavong P, Lorphacan L, Soundala P, Hongvanthong B, Ohta N, Brey PT, Kano S. The application of the LAMP method to Opisthorchis viverrini and Schistosoma mekongi infections in Champasak Province, southern Lao PDR. 10th National Health Research Forum, Savannakhet province, Lao PDR, October 27th–28th, 2016.
Poster presentation:
Pongvongsa T, Nonaka D, Iwagami M, Xangsayarath P, Nishimoto F, Khattignavong P, Soundala P, Lorphacan L, Hongvanthong B, Brey PT, Kano S, Kobayashi J. Malaria among foreign migrant workers in Savannakhet Province, Lao PDR. 10th National Health Research Forum, Savannakhet province, Lao PDR, October 27th–28th, 2016.
Iwagami M, Khattignavong P, Soundala P, Lorphachan L, Keomalaphet S, Xangsayarath P, Hongvanthong B, Brey PT, Kano S. Current Malaria Situation in five southern provinces, Lao PDR—A study by the SATREPS Project. Joint International Tropical Medicine Meeting 2016, Bangkok, Thailand, December 7th–9th, 2016.
Acknowledgement / Funding:
We wish to thank Dr. Bouasy HONGVANTHONG, Director of the Center of Malariology, Parasitology and Entomology (CMPE), Ministry of Health, Lao PDR, and Project director of SATREPS for his kind support of this project. We thank Dr. Tiengkham PONGVONGSA, Malaria Station Director, Savannakhet Provincial Health Department, Lao PDR. We also thank the staff of the CMPE, the NIOPH, Provincial Health Offices, Provincial Hospitals, District Hospitals, and Health Centers, Lao Ministry of Health, for supporting our field surveys and training courses.
This work is supported by the SATREPS (Science and Technology Research Partnership for Sustainable Development) project from the Japan International Cooperation Agency (JICA) and the Japan Agency for Medical Research and Development (AMED).

References:
1. Ashley E.A., Dhorda M., Fairhurst R.M., Amaratunga C., Lim P., Suon S., et al. Spread of artemisinin resistance in Plasmodium falciparum malaria. New England Journal of Medicine, 371: 411–423, 2014.
2. Ménard D., Khim N., Beghain J., Adegnika A., Alam M., Amodu O., et al. A Worldwide Map of Plasmodium falciparum Arteminisin Resistance. New England Journal of Medicine, 374:2453–2464, 2016.
3. Howes R.E., Dewi M., Piel F.B., Monteiro W.M., Battle K.E., Messina J.P., et al. Spatial distribution of G6PD deficiency variants across malaria-endemic regions, Malaria Journal, 12: 418, 2013.
4. WHO Working Group, Glucose-6-phosphate dehydrogenase deficiency. Bulletin of the World Health Organization, 67:601–611, 1989.
5. Matsuoka H., Thuanb D.T.V., Thienb H.V., Kanbec T., Jallohc A., Hiraia M., et al. Seven Different Glucose-6-phosphate Dehydrogenase Variants Including a New Variant Distributed in Lam Dong Province in Southern Vietnam. Acta Medica Okayama, 61:213–219, 2007.
6. Matsuoka H., Wang J., Hirai M., Arai M., Yoshida S., Kobayashi T., et al. Glucose-6-phosphate dehydrogenase (G6PD) mutations in Myanmar: G6PD Mahidol (487G>A) is the most common variant in the Myanmar population. Journal of Human Genetics, 49:544–547, 2004.












