Mapping of Vectors and Reservoir Background Hosts in Laos (Tick Map 5)
1 Project Coordinator:
Project Coordinator:
• Dr. Khamsing Vongphayloth, Research entomologist, Institut Pasteur du Laos
• Dr. Jodi Fiorenzano, Chief of Entomological Sciences, U.S. Naval Medical Research Center – Asia, Sembawang, Singapore
• Dr. Paul Brey, Director, Institut Pasteur du Laos, Vientiane, Lao PDR
Staff members:
• Khaithong Lakeomany, Technician entomologist, Institut Pasteur du Laos
• Nothasine Phommavanh, Technician entomologist, Institut Pasteur du Laos
• Veaky Vungkyly, Technician entomologist, Institut Pasteur du Laos
Partners
• The Lao-Oxford University-Mahosot Hospital-Wellcome Trust Research Unit (LOMWRU)
• The Pathogen Discovery Laboratory, IP-Paris-France Funded by the U.S. Naval Medical Research Center-Asia (NMRC-A) in support of the Department of Defense Global Emerging Infections Surveillance and ResponseSystem (DoD-GEIS)
Period of Project: 2021-2022
Background
Vector-borne diseases constitute a significant infectious disease risk for deployed military personnel and for local populations. In Laos, definitive diagnosis is often not available for vector-borne illnesses, so the infectious diseases which are a threat to military and civilian populations are not well-defined. In order to identify common and emerging vector-borne pathogens in Laos, NAMRU-2 Singapore (SG) has established a study to assess the distribution and infection potential of vectors (including ticks and associated arthropods). In this study, tick and associated arthropod vectors will be surveyed from the environment and their associated hosts to provide biological specimens for diagnostic purposes. The samples will be transported to the Institut du Pasteur (IPL) laboratory in Vientiane, where a wide range of diagnostic tests can be performed to identify both the vector and pathogens with which they may be infected. In order to understand the infectious disease threats in a range of environments in Laos, IPL must collect and screen specimens from at least 2 sites and 2 provinces within Laos.
Strategic objective
• Collect up to 10,000 ticks and associated arthropod specimens from study areas according to the study protocol and deliver those samples to the IPL laboratory in Vientiane.
Specific objectives
• Survey and modern identification of indigenous tick and associated arthropod species.
• Collection, ID, extraction of vector DNA for submission and development of regional repository. This will provide a valuable resource for downstream modern molecular analysis and genotyping. • Building of local capacities and competencies.
Major Milestones
• (Q1) Develop protocol for collection, purchase equipment/supplies required for entomological collections, DNA/RNA extraction, and molecular characterization.
• (Q2/Q3) Collection (at least 2 field missions), identification, extraction, and pathogen screening.
• (Q4) Pathogen screening, present results and submit final report.
Methodology and progressing results
Field site and times
Our first field mission of 12 days was conducted between 16 and 28 January 2022 in Luangnamptha province. Five sites of Viengphoukha district, Luangnamtha province were selected for sampling. Cage traps were set in two villages, namely Nam-karb (20.705665°, 101.006957°) and Nam-eng (20.721118°, 101.143051°). Tick dragging was conducted in Nam-karb village and Nam Ha national protected area (20.865785°, 101.311191°). Additional tick collection was carried out by examining domestic animals (cows) in Nam-karb, Nam-eng, Namsing (20.766984°, 101.208272°), and Prang villages (20.782823°, 101.236057°).
Our second field mission was conducted between 3 and 15 May 2022 at the same sites as our first field mission. Two more villages of Namtha district were added for tick collection. Cage traps were set in two villages: Prang village, Viengphoukha district; and Khuasoung village (20.802732°, 101.250440°), Namtha district.
Our third field mission was conducted between 17 and 29 July 2022 Sing district, Luangnamptha province. Ectoparasites were surveyed from 4 villages: Chiengchai (21.189537°, 101.151660°), Chorm (21.236294°, 101.154985°), Na Mai (21.221399°, 101.148957°), and Tin Thad (21.157852°, 101.164764°).
Field collection procedure
Tick dragging/flagging: Tick dragnets were swept/ dragged along the forest ground at approximately 1–2 m intervals before being examined for ticks. Ticks were removed from the sheets using forceps, then transferred to 1.5 ml labeled cryotubes, and stored -20°C. Our dragnet collecting was carried out in all three sites.
Small Mammal Trapping: rodent traps were set for 6 nights in each two missions in Viengphoukha district and 6 nights in Sing district. In each study site, 50 traps (baited with bananas, sticky rice, or dried fish/meat) were placed in the transect format according to the topography. All rodents were released after checking for ectoparasites.
Additional ectoparasite collection was carried out by examining domestic animals. The animal owners were asked to help to examine their animals. Once ticks attached on animals were found, they were collected by direct hand removal with forceps.
All ectoparasite samples were stored in -20°C in the field and transported to Vientiane laboratory (IPL) using dry ice.
Laboratory work
Sample identification and preparation
Ticks were identified and grouped under microscopes in cooling conditions (on ice packs) by using reference determination from Dr. Richard G. Robbins of the US Armed Forces Pest Management Board (AFPMB), together with related references from Southeast Asia, Japan, Korea, the Ryukyu Islands (Yamaguti, Tipton et al. 1972), L. E. Robinson keys for genus Amblyomma (Nuttall, Cooper et al.), and keys from Thailand (Tanskull and Inlao 1989) for adult Haemaphysalis ticks. As there are no morphological identification keys available for pre-imago forms, all larval and nymph stages were grouped into genus. After tick identification and pooling, all information was registered with the Pathogen Asset Control System (PACS) software and all tick samples were stored at −80°C in IPL for further analysis.
Chigger mites from rodents were mounted on slides using PVA mounting medium. Mite samples were identified using compound microscopes to genus level by referring to a published taxonomic key of Nadchatram & Dohany, 1974.
Sample preparation and RNA/DNA extraction
Specimens were placed in a 1.5 ml vial containing 1 ml of 1X cold Phosphate Buffered Saline (PBS) and Lysing Matrix A zirconium beads (MP Biomedicals). Tick pools were homogenized for 10 min at a vibration frequency of 25/s in a TissueLyser II system (Qiagen). After grinding, beads and tissues were spun down by centrifugation for 5 min at 3000 rpm. To obtain total nucleic acid (both DNA and RNA) for bacterial and viral detection by polymerase chain reaction (PCR), 100 μl of each pool was extracted and purified by using NucleoSpin® 8 Virus extraction kit following the manufacturer’s protocol. The remaining 400 μl of each pool was stored at –80°C for future pathogen isolation.
Arboviral screening at IPL
Phleboviruses and flaviviruses were screened by means of conventional nested RT-PCR as previously described (Sanchez-Seco, Rosario et al. 2003; Sanchez-Seco, Rosario et al. 2005).
Bacterial screening
The bacterial screening was carried out in collaboration with the Lao-Oxford University-Mahosot Hospital- Wellcome Trust Research Unit (LOMWRU), based at Mahosot Hospital, Vientiane. To investigate the occurrence of Rickettsia spp. in ticks, a molecular screening approach targeting the 17kDa gene was taken (Jiang et al. 2004). The presence of Anaplasma spp. and Coxiella was also investigated.
Next generation Sequencing at IPP
A total of 24 big pools containing 259 mini pools (1,707 ticks) were sent to IPP for more deep sequencing.
Results
Number, species abundance and composition of ticks and other ectoparasites
A total of 2,748 samples of ticks and other ectoparasites were collected during our course of study, of which 729,1,146 and 873 samples were collected from first, second and third missions respectively (Table 1 for more detail).
Six species belonging to 4 genera of ticks were collected from dragging and animals including A. testudinarium, H. hystricis, I. spp., R. haemaphysaloides, R. microplus, and R. sanguineus. Trombiculidae and Haematopinidae ectoparasites were also collected from only rats and animals (Table 2 below for more detail).
Table 1: Number of ticks and other ectoparasites collected according to methods and location.
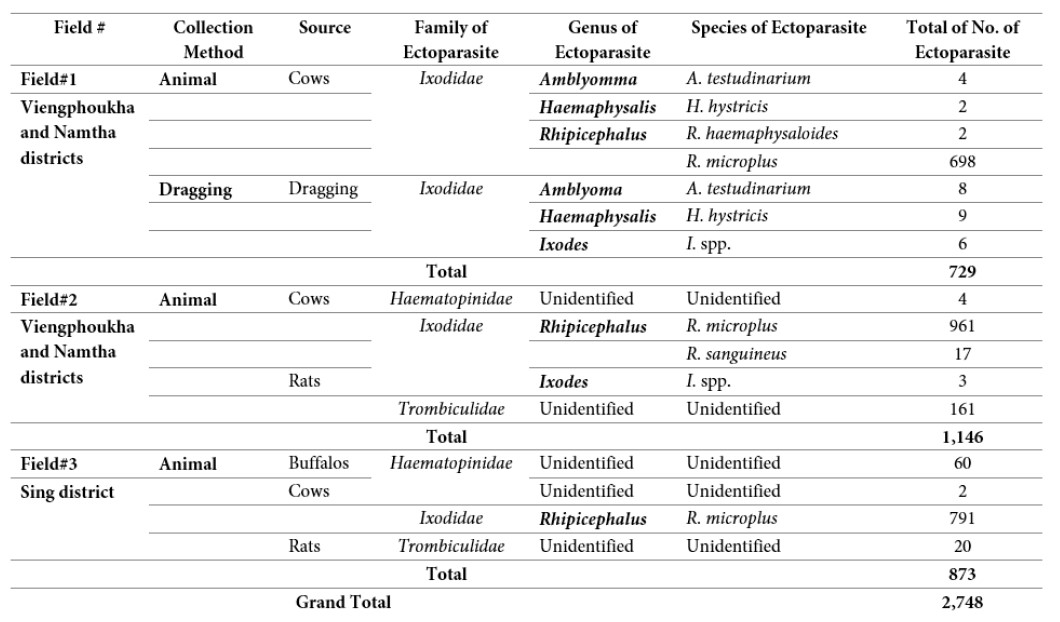
Table 2: Species and number of ticks and other ectoparasites collected by different methods.

Preliminary results on pathogens associated with ticks
Arboviral screening
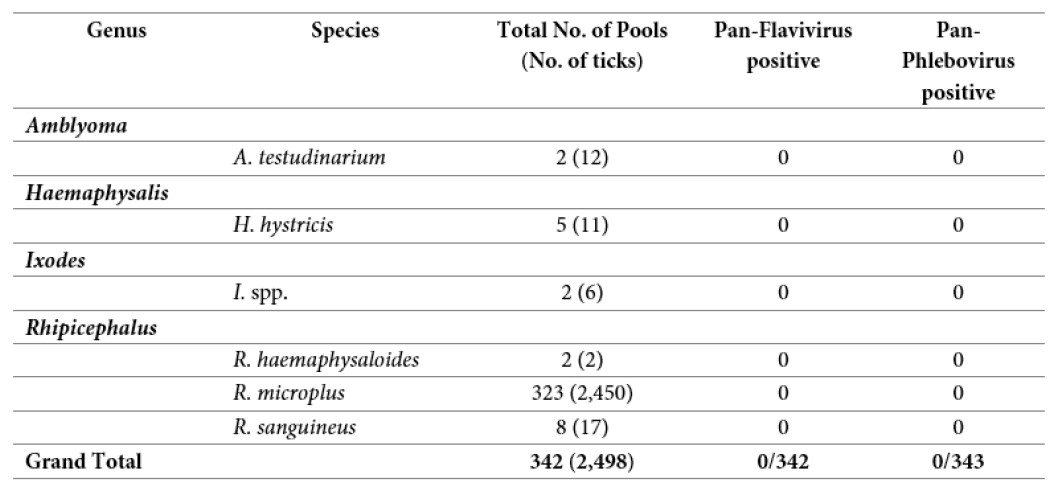
A total of 342 pools (2,498 tick samples) were screened for Pan-Phlebovirus and Pan-Flavivirus by conventional nested RT-PCR. None of them were positive (Table 3).
Table 3: Results of Pan-Flavivirus and Pan-Phlebovirus RT-PCR screening
Bacterial screening
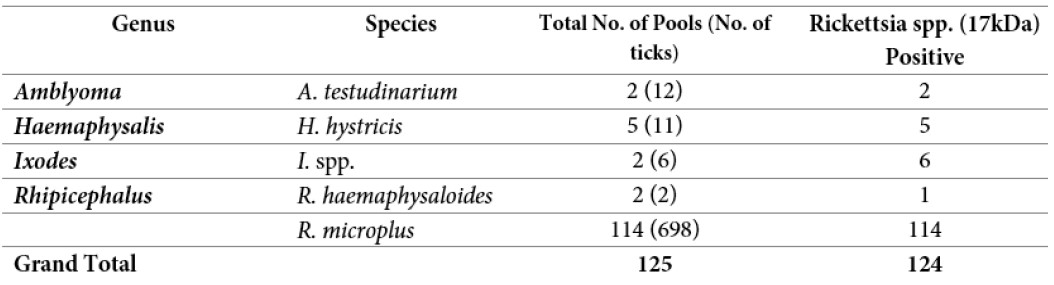
So far, a total of 125 pools containing 729 ticks have been screened for the presence of Rickettsia spp. at Lao-Oxford- Mahosot Hospital-Wellcome Trust Research Unit (LOMWRU). A total of 124 pools were positive for Rickettsia spp. (17kDa). The Rickettsia screening is ongoing.
Table 4: Rickettsial screening from ticks
Next generation Sequencing at IPP
A total 24 big pools containing 259 mini pools (1,707 ticks) were sent to IPP in August 2022 for more deep sequencing. Our IPP partner will conduct NGS on the 24 big pools. The results of NGS will be submitted by the end of November 2022.
Problems Identified during our project and Follow-up Actions
• Our project continues to move forward with no currently observed problems or major obstacles. We have finished all of our field collections as planned.
• Our partner, LOMWRU, will continue to try to identify the Rickettsia species.
• Our partner at IPP will continue to work on deep sequencing of samples that we sent in August 2022 for Arbovirus.
• We propose to continue to study ticks in Laos for both tick identification and associated pathogens.
References
Jiang J., Chan T., Temenek J.J., Dasch G.A., Ching W. and Richards A.L. (2004). Development of a Quantitative Real time Polymerase Chain Reaction Assay Specific for Orentia Tsutsugamushi. American Journal of Tropical Medicine and Hygiene, 70(4): 351-356.
Nadchatram, M. and A. L. Dohany. 1974. A pictorial key to the subfamilies, genera and subgenera of Southeast Asian chiggers (Acari, Prostigmata, Trombiculidae). Bulletin from the Institute for Medical Research Federation of Malaysia, 16: 1–67.
Nuttall, G. H. F., W. F. Cooper, C. Warburton, L. E. Robinson, and D. R. Arthur. 1926. Ticks: pt. IV. The genus Amblyomma. Cambridge University Press.
Sanchez-Seco, M.P., et al., Detection and identification of Toscana and other phleboviruses by RT-nested-PCR assays with degenerated primers. J Med Virol, 2003. 71(1): p. 140-9.
Tanskull, P. and I. Inlao. 1989. Keys to the adult ticks of Haemaphysalis Koch, 1844, in Thailand with notes on changes in taxonomy (Acari: Ixodoidea: Ixodidae). J Med Entomol 26(6): 573–600.
Yamaguti N., V. J. Tipton, H. L. Keegan, and S. Toshioka (1971). Ticks of Japan, Korea, and the Ryukyu islands. Brigham Young University Science Bulletin, Biological Series 15(1):1.










