Immunity levels to poliovirus in Lao children and adults before the vaccine-derived poliovirus outbreak: a retrospective study
 Project coordinators: Maude Pauly, Claude Muller
Project coordinators: Maude Pauly, Claude Muller
Staff Members: Phonethipsavanh Nouanthong
Background
Large-scale vaccination campaigns eliminated wild poliovirus (WPV) in most countries of the world and WPV remains endemic only in Pakistan, Nigeria and Afghanistan. In the Lao People’s Democratic Republic (Lao PDR), oral polio vaccine (OPV) was introduced in the early eighties, and vaccination was expanded to the whole country in the nineties. In 1996, the last case of WPV was reported in Lao PDR and in 2000, the Western Pacific Region, including Lao PDR, was certified polio-free.
Despite its ground-breaking role in the eradication of poliovirus (PV), OPV (containing live attenuated PV strains) has some important drawbacks. A small proportion of vaccinees develop vaccine-associated paralytic poliomyelitis (VAPP).
In countries with suboptimal vaccination coverage levels and weak acute flaccid paralysis (AFP) surveillance, excreted vaccine-virus can replicate and circulate for a prolonged time. Within less than a year of circulation, vaccine viruses may accumulate genetic mutations and neurovirulent vaccine-derived PV (VDPV) may emerge.
At the end of 2015, VDPV type 1 strains emerged in Lao PDR and caused paralysis in 11 individuals (last case in January 2016) in the three neighboring provinces Bolikhamxay, Xaisomboun and Vientiane Province (see also Fig 1). Most of the affected were male; 4 were below the age of 18 months, 4 between 4 and 15 years and 2 were in their forties. Only a one-year old child had completed the three OPV vaccinations. The others had only one or no dose of OPV. Virtually all belonged to the ethnic group of the Hmong. Circulation of the VDPV (cVDPV) was confirmed by epidemiological investigations and molecular analyses. By the beginning of 2016, the outbreak was declared a public health emergency by the Prime Minister and large vaccination campaigns with trivalent OPV were launched and surveillance was intensified throughout the country.
To reduce the risk of cVDPV emergence and in compliance with the Polio End Game Strategy, a switch from trivalent (containing type 1, 2 and 3 vaccine strains) to bivalent (containing type 1 and 3 vaccine strains) OPV was globally implemented in 2016. In Lao PDR, the new recommendations, introduction of bivalent OPV and of inactivated vaccine (IPV) as an adjunct to routine immunization with OPV will soon be implemented.
Currently, PV surveillance relies mainly on the reporting of AFP and in Lao PDR, reported rates of AFP reached almost the recommended minimum required for PV-free countries (i.e. one per 100 000 children below 15 years of age). However, PV surveillance remains challenging as the silent circulation of WPV and VDPV is frequent.
Not enough is known in Lao PDR about immunity levels against PV to understand the reasons for the recent VDPV outbreak. We investigated five large cohorts with different epidemiological, demographic, exposure and vaccination histories in order to provide important public health information about polio epidemiology.
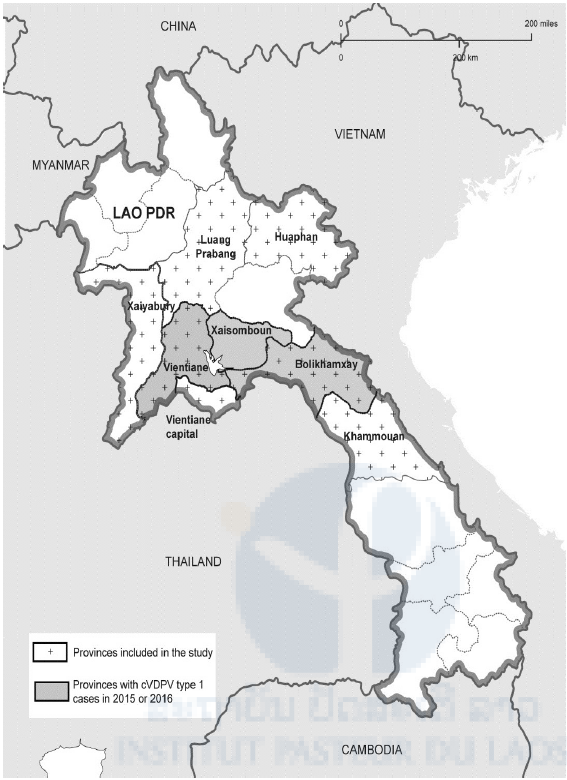
Fig 1. Lao provinces. Provinces affected by the cVDPV type 1 outbreak are represented in grey and provinces included in the study are dotted.
Results
Seroprevalence in children (Cohorts 1-3)
97.6% of the children with full vaccination records and aged less than 3.5 years had IgG antibodies against PV by ELISA (Cohort 1). Neither sex or age, nor birthplace or nutritional status influenced PV immune status. However, seropositivity rates differed significantly between the 3 provinces (χ2=14.7, df=2, p<0.01; Table 1).
An anti-PV antibody seroprevalence of 98.8% was determined in children aged less than 5 years from remote districts in Huaphan (Cohort 2).
Neither age or sex, nor distance to next health care facility had a significant effect on PV immune status (Table 1). In another cohort of children with unknown vaccination status from less marginalized communities (Cohort 3), antibody seroprevalence was somewhat lower with 92.3%. Similar to what was found for cohort 1, significantly lower immunity rates were also found for this cohort in Bolikhamxay (84.1%) than in the two other provinces (Luang Prabang: 95.4%, OR=3.9, 95%CI=1.4- 10.6, p=0.01; Vientiane: 96.3%, OR=4.9, 95%CI=1.4- 18, p=0.007). Children aged less than 1 year were significantly more likely to have anti-PV antibodies than children aged 5 to 9 years (97.6% versus 88.6%, p=0.007) and anti-PV antibody levels were negatively correlated with age (Pearson’s r=-0.4; 95%CI=-0.5 and -0.3; t=-7.1, df=318, p<0.001).
Seroprevalence in adults (Cohorts 4 and 5)
81.7% of the blood donors (Cohort 4) had antibodies against PV by ELISA. Seroprevalence rates ranged from 81.1 to 83.3% in the 4 provinces with no significant differences. Participants born after the introduction of OPV into the national immunization program in 1979 were significantly more likely to be seropositive: only 69.2% of the birth cohort 1958-1977 were seropositive, compared to 82.7 and 85.1% of the younger birth cohorts (1978-1988: OR=2.1, 95%CI=1.1-4, p=0.028; 1989-1998: OR=2.5, 95%CI=1.4-4.6, p=0.003) (Table 1, Fig 2A). Anti-PV antibody levels correlated negatively with age (Pearson’s r=-0.1; 95%CI=-0.2 and 0; t=-3.6, df=526, p= 0.001) (Table 1, Fig 2B). Mean antibody levels were significantly lower in blood donors (47.6, 95%CI=18.2- 69.6) than in the fully vaccinated children (79.1; 95%CI=47.7-79.1) (t=-14.3, df=1031.5, p-value<0.001; Fig 2C).
Overall, a lower seroprevalence was determined for HCW (Cohort 5) than for blood donors: only 71.9% of the HCW had antibodies against PV by ELISA. Also in this adult cohort, the antibody seroprevalence decreased with age from 79.7% (the birth year 1989-1998) to 74.3% (the birth year 1978-1988), to 69% (the birth year 1958-1977), but between the youngest and the oldest cohort, there was only a trend to significance (p=0.067).
Anti-PV antibody levels were negatively correlated with age (Pearson’s r=-0.2; 95%CI=-0.2 and -0.1; t=-2.9, df=698, p-value=0.004). The other recorded risk factors were not significant (Table 1).
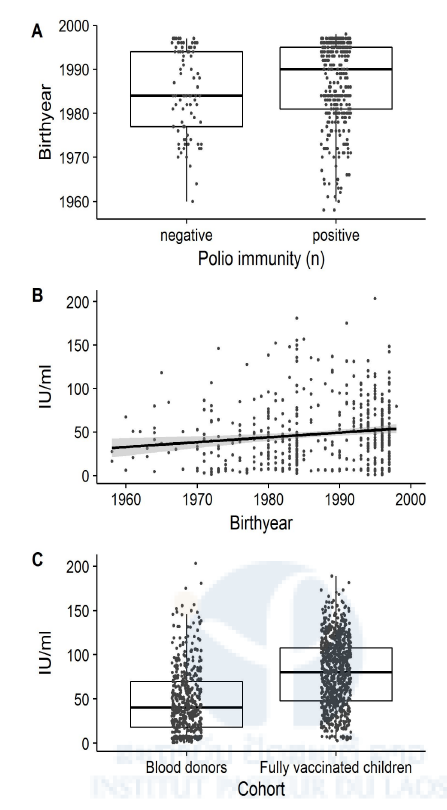
Fig 2. Age-related differences in poliovirus immunity as determined by ELISA
In our adult cohorts, we found that only 81.7% of the Lao blood donors and 71.9% of the HCW had anti-PV antibodies by ELISA. There was no significant difference between blood donors from different provinces and between HCW from central, provincial or district hospitals or with different clinical and non-clinical positions. In both adult cohorts, older participants and particularly those born before the introduction of OPV in the country had significantly lower anti-PV antibody levels and, were significantly less likely to be seropositive (Figs 2A and B) than younger adults or the children of Cohort 1 and 2. Adults born before the eighties did not undergo routine vaccination and were also less likely to participate in SIAs. The significant age-dependent decrease in total anti-PV antibodies determined by ELISA among all adult cohorts could thus be a reflection of the lower vaccination coverage in this age group, or of the waning immunity after vaccination or exposure to VDPV or WPV. This may explain why four of the eleven reported paralytic cases occurred among individuals above the age of 14 years.
Thus, our seroprevalence results are in line with what was observed during the outbreak. However, a limitation of our cohorts was that for ethical reasons we were unable to collect explicit information on ethnicity, while ethnic minority communities were most affected by the outbreak. These and adult risk groups such as HCWs, and regions with weak vaccination programs should be primarily targeted by future supplementary vaccination activities.
These data have been published as a manuscript in Plos One and reported to the National Immunization Technical Advisory Group and the National Immunization Programme in the form of a policy brief. The data were also presented as a poster at the Global Vaccine Research Forum in Bangkok, 2018.
Table 1. Association between socioeconomic characteristics and anti-polio IgG seropositivity as determined by ELISA for each study cohort.
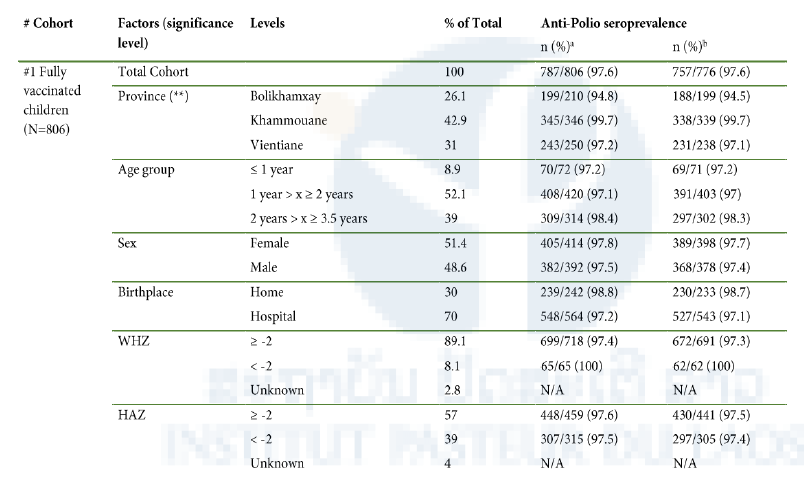
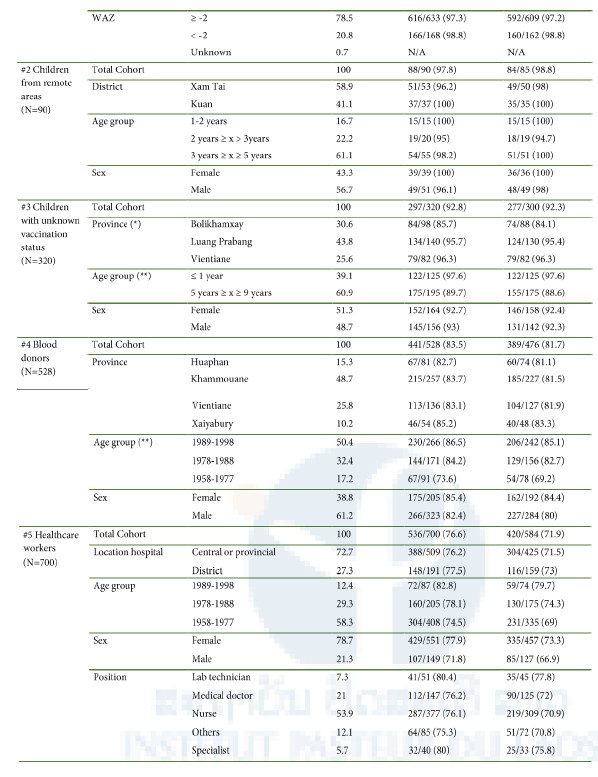
aComplete dataset, borderlines being considered positive
bDataset without borderline samples
*significant effect on anti-poliovirus antibody seroprevalence (p-value between 0.05 and 0.01)
**highly significant effect on anti-poliovirus antibody seroprevalence (p<0.01)









