Efficacy of alternative larvicides for dengue vector control in semi-field trial
Project leader: Dr. Sebastien Marcombe, Dr. Ian Sutherland and Dr. Paul Brey
Member of staff: Khamsing Vongpayloth (Scientist) Noy Khounsombat, (Laboratory technician) Nothasine Phommavanh (Laboratory technician)
Bounma Xayasouk (Mosquito insectary technician)

Background
To provide information on alternatives to temephos for vector control operations in the Lao PDR, we studied the resistance levels of Aedes aegypti mosquitoes from Vientiane to Bacillus thuringiensis var israeliensis (Bti, bio-insecticide) and the spinosad (naturalyte), each of which has a different mode of action. We also tested the efficacy of the insect growth regulator diflubenzuron (benzoylurea) that acts by disrupting chitin synthesis and deposition (Matsumura 2010). This IGR showed a promising efficacy against several mosquito species, especially Aedes aegypti. Bti has desirable properties for mosquito control because of its fast killing effect, a good toxicological profile (Lacey 2007), and the absence of cross-resistance with conventionally used pesticides. All of the above insecticides are recommended by the World Health Organization (WHO) for use for vector control in drinking water sources and containers and may be used routinely by mosquito control services (WHO 2004, 2008, 2010).
The purpose of this study was to characterize the resistance status of Ae. aegypti larvae from Vientiane, Lao PDR, to temephos, Bti, and diflubenzuron and to assess the efficacies and residual activities of these insecticides under simulated conditions.
Materials and methods
The trial was carried out in Vientiane, Lao PDR, on the premises of the Institut Pasteur du Laos. The tents and the plastic containers were installed in early October and the experiment started on the 8th of October (Figure 9). The effects of temephos (1mg/L), diflubenzuron (0.25mg/L), and Bti (8mg/L) formulations were evaluated and compared against Ae. aegypti larvae (IPL strain). Blue plastic containers with a capacity of 200 liters were used because they are widely used for water storage in Vientiane City and have been shown to be an important productive breeding habitat for Ae. aegypti. These drums were filled with 175 liters of domestic water and covered with a mosquito net to prevent oviposition by wild female mosquitoes in the area and to prevent the deposition of debris. The containers were placed under a shelter to prevent direct exposure to rain and sunlight (Figure 9). All the insecticides were tested at the dosage recommended by the WHO or the manufacturers for the control of mosquito larvae. Twelve containers (three replicates per insecticide) were allocated to insecticides at random. Three were left untreated and used as a control. Groups of 100 third-instar larvae of the F1 generation of the Institut Pasteur du Laos strain (IPL strain collected in sentinel containers at the Institute) were added to each container with one gram of food (dry cat food) at time 0, and then every 10 days. The containers were replenished every 10 days to maintain the initial level of water. Emerging adults were collected from each container by using electric aspirators and then stored at −80°C. Temperature and pH were checked every 10 days with a portable tester to detect any differences between replicate and/or treatments. External temperature and humidity were recorded by using a meteorological unit. Emergence inhibition rates (% EI) and 95% CIs were calculated for the average of the three replicates per insecticide according to the formula: % EI = ((C – T)/C) × 100 where C is the emergence in the control and T is the emergence in the treated container at the same time period. For each formulation, curves were presented until the % EI decreased to <80%, which corresponded to the threshold generally considered for reapplication of the treatment.
The susceptibility status of the IPL strain against Bti and diflubenzuron was measured following the same protocol described above.

Figure 9. Semi field trial at the Pasteur Institute of Laos. Efficacy of conventional versus alternative insecticide formulations used for vector control are tested.
Results
Results of the larval bioassays on the IPL strain (Vientiane capital) are shown in Table 7. The result showed that the IPL strain was fully susceptible to the insecticides Bti and Diflubenzuron showing that these insecticides are good candidates for vector control in the Lao PDR.
Table 7: Resistance status of Aedes aegypti (IPL strain) against Bti and Diflubenzuron

Resistant ratios = LC50 of wild strain / LC50 of USDA susceptible reference strain.
The results of the semi-field experiment are presented in Figure 10. Twenty-three weeks after the treatments, the average percentage of emergence in the containers treated with Bti was more than 20%, which is the limit recommended for a new treatment in field conditions. This threshold was exceeded in the containers treated with diflubenzuron after twenty-four weeks, while the temephos treatment showed 96% efficacy (4% emergence) after twenty-eight weeks. One should note the dramatic decrease of emergence in the control containers dropping from 80% to 40% between T110 and T150 (Figure 10). This corresponds to an increasing temperature inside the containers during the same period. During this period, the temperatures rose from 22o C to almost 30o C inside the containers. This event may have consequences also on the treated containers, delaying the increase of emergence for a few weeks.
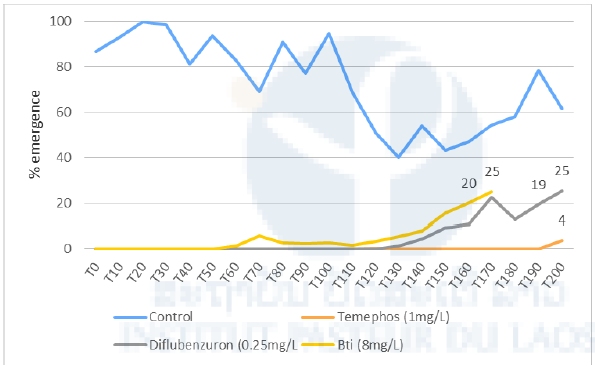
Figure 10. Percentage emergence of Ae. aegypti in the control and treated containers
Discussion and Conclusions
The semi-field trial assays showed that after 22 weeks, all the insecticides tested (i.e. Bti, temephos, and diflubenzuron) have remained above 80% Emergence Inhibition (EI), i.e. the acceptable threshold for a larvicide according to WHOPES (WHO, 2005). Below 80% EI, a new treatment is recommended. Our results indicated a good residual efficacy of these three insecticide formulations against Aedes aegypti. However, it has already been pointed out that the performance of larvicides in well-controlled conditions has to be taken cautiously because of the absence of direct exposure to rain, sunlight, and organic matter that could lead to an over-estimation of the residual activity of an insecticide. Simulated-field experiments represent a useful method for screening new insecticides and/or to select dosages for field application, but they cannot be used to predict the performance of formulated products under real conditions. Despite the relatively good residual activity of the temephos formulation, we should take into account that several populations presented moderate levels of temephos resistance (i.e. RR95>2). Temephos insecticide is the active ingredient of the Abate formulation, the larvicide used for dengue vector control in the Lao PDR. Those levels of insecticide resistance indicate that a selection pressure occurred in these mosquito populations. This emphasizes the need to find a substitute for the Abate formulation to avoid the development of resistance and make future public health operations useless. Furthermore, Abate is posing environmental issues and was banned in the European Union in 2009. The future use of more environmentally friendly products such as Bti and diflubenzuron is recommended, especially knowing that no cross-resistance between these insecticides and temephos has been reported.
The adult bioassays showed that most of the Ae. aegypti adult populations were highly resistant to permethrin and DDT, but susceptible to deltamethrin. Permethrin and deltamethrin belong to the pyrethroid family and have been used in the Lao PDR for adult control since the nineties to fight against malaria and dengue. Both have been used in space-spraying applications or in treated bed nets and can be found in household aerosol sprays. This long and intense insecticide use has probably led to the permethrin resistance and deltamethrin resistance is likely to follow soon his long and intense insecticide use has probably led to the permethrin resistance and deltamethrin resistance is likely to follow soon (moderate levels of resistance in larvae, Table 2). Despite the fact that DDT was banned in 1989 in the Lao PDR, strong levels of resistance have been observed.
This could be explained by the well-known cross-resistance occurring between pyrethroids and organochlorine (DDT). The use of malathion since the nineties for dengue control certainly explains the high resistance levels in Aedes populations in the Lao PDR.
We investigated the presence and the frequency of two well-known kdr mutations (V1016G and F1534C) in 11 field-caught populations of Aedes aegypti in the Lao PDR. The results showed that the 1016G and 1534C kdr alleles are present in the Lao PDR but at various frequencies. The 1534C was found at high prevalence (>0.6 except in one population) whereas the 1016G was found at low prevalence (<0.4). In our study no correlation was found between the presence of mutant alleles and the survival rates of mosquitoes to DDT and Permethrin. This can be explained by significant linkage disequilibrium (as discussed above) and/or by the presence of additional mechanisms (metabolic) playing a greater role in resistance than the kdr. Brooke and colleagues (2008) have already showed early showed that resistance could be multigenic, and the kdr genotype might not fully explain all the variance in phenotype. This was confirmed recently by a study published in Genome Research that showed that none of the V1016I/G kdr mutations previously associated with pyrethroid resistance were retained as best candidate markers (Faucon et al., 2015). Contradictory findings on the role of the kdr mutations in pyrethroid resistance were previously reported (Saavedra-Rodriguez et al., 2007; Donelly et al., 2009). Clearly further validation studies are needed to address the exact role of the various kdr mutations in DDT and pyrethroid resistance.
We investigated the role of the detoxification enzymes in the resistance in five resistant populations from Vientiane (2 populations), Luang Prabang, Saravane and Xayabury. Results showed significant differences in detoxification enzyme activities/quantities compared to the susceptible reference strain (USDA) suggesting the involvement of metabolic based resistance mechanisms. Our toxicological data with the use of synergists supported these results. The efficacy of malathion and permethrin against the populations tested was restored after the mosquitoes were exposed to inhibitors of detoxification enzymes. Esterases based-resistance mechanism is a major mechanism for organophosphate resistance in insects (Hemingway and Karunaratne, 1998). Several examples of Ae. aegypti resistance to organophosphates worldwide linked to elevated carboxylesterases activities have been described (Rodriguez et al., 2001; Wirth et al., 1999). Our toxicological and biochemical data confirmed these observations. Among detoxification enzymes, P450s have been shown to play a major role in pyrethroid resistance in insects (Brogdon et al., References 1998; Hemingway et al., 2004; Ranson & Hemingway, 2005).
In the Caribbean, Marcombe et al. (2009) suggested the involvement of P450s in the reduced efficacy of deltamethrin space-spray operations. Elevated GST levels have also been frequently associated with insect resistance to insecticides such as DDT and pyrethroids (Vontas et al., 2001; Enayati et al., 2005; Lumjuan et al., 2005). Our toxicological and biochemical data support the role of P450s and GSTs in insecticide resistance in the Lao PDR.
Better understanding of the levels and mechanisms of insecticide resistance will help to improve the surveillance, the management, and the control of insecticide-resistant dengue vectors in the Lao PDR. The development of new tools such as molecuar biomarkers (i.e. gene Copy Number Variations [CNVs], Single Nucleotide Polymorphisms [SNPs] and differentially expressed genes [DE genes]; Faucon et al. 2015) is urgently needed to fight against dengue and to contribute to a fastest, costless and more efficacious control operations in resistant mosquito population areas..
+ It is important to maintain a regular monitoring of the levels of insecticide resistance in the different provinces of the Lao PDR to detect the development of insecticide resistance in the dengue vectors
+ We recommend the use of alternative insecticides where insecticide resistance is detected
+ We recommend to start using new insecticides in rotation (i.e. a different insecticide each year) to avoid or to manage the development of insecticide resistance in the larval and adult populations of the dengue vectors in the Lao PDR
Partners
+ U.S. Naval Medical Research Center – Asia
+ National Center of Malariology, Parasitology and Entomology (CMPE), Vientiane, Lao PDR
+ Kasetsart University, Bangkok, Thailand
+ Institut de Recherche pour le Développement, MIVEGEC (UM1-CNRS 5290-IRD 224)
Financial support
The ARBOVEC project was initiated thanks to the research grant from U.S. Naval Medical Research Center – Asia. Agreement No.: W911QY-14-2-0003
References
1. Brengues C, Hawkes NJ, Chandre F, McCarroll L, Duchon S, et al. (2003) Pyrethroid and DDT cross-resistance in Aedes aegypti is correlated with novel mutations in the voltage-gated sodium channel gene. Medical and Veterinary Entomology 17: 87-94
2. Brogdon WG, McAllister JC: Insecticide resistance and vector control. Emerg Infectious Diseases 1998, 4 (4):605-613.
3. Brooke, BD (2008) Kdr: can a single mutation produce an entire insecticide resistance phenotype? Trans. R. Soc. Trop. Med. Hyg. 102, 524–525
4. Donnelly MJ, Corbel V, Weetman D, Wilding CS, Williamson MS, Black WC 4th. Does kdr genotype predict insecticide-resistance phenotype in mosquitoes? Trends Parasitol. 2009. 25(5):213-9.
5. Enayati AA, Ranson H, Hemingway J: Insect glutathione transferases and insecticide resistance. Insect Mol Biol 2005, 14(1):3-8.
6. Faucon F, Dusfour I, Gaude T, et al. Identifying genomic changes associated with insecticide resistance in the dengue mosquito Aedes aegypti by deep targeted sequencing. Genome Research. 2015;25(9):1347-1359.
7. Harris AF, Rajatileka S, Ranson H: Pyrethroid resistance in Aedes aegypti from Grand Cayman. Am J Trop Med Hyg 2010, 83:277-284.
8. Hemingway J, Field L, Vontas J (2002) An overview of insecticide resistance. Science 298: 96-97.
9. Hemingway J, Hawkes NJ, McCarroll L, Ranson H (2004) The molecular basis of insecticide resistance in mosquitoes. Insect Biochemistry and Molecular Biology 34: 653-665.
10. Hemingway J, Karunaratne SH (1998) Mosquito carboxylesterases: a review of the molecular biology and biochemistry of a major insecticide resistance mechanism. Medical and Veterinary Entomology 12: 1-12.
11. Kawada H, Higa Y, Komagata O, Kasai S, Tomita T, Thi Yen N, Loan LL, Sanchez RA, Takagi M: Widespread distribution of a newly found point mutation in voltage-gated sodium channel in pyrethroid-resistant Aedes aegypti populations in Vietnam. PLoS Negl Trop Dis 2009, 3:e527.
12. Kawada H, Oo SZ, Thaung S, Kawashima E, Maung YN, Thu HM, Thant KZ, Minakawa N: Co-occurrence of point mutations in the voltage-gated sodium channel of pyrethroid-resistant Aedes aegypti populations in Myanmar. PLoS Negl Trop Dis 2014, 8:e3032.
13. Lacey LA, 2007. Bacillus thuringiensis serovariety israelensis and Bacillus sphaericus for mosquito control. J Am Mosq Control Assoc 23: 133 – 163.
14. Lumjuan N, McCarroll L, Prapanthadara LA, Hemingway J, Ranson H: Elevated activity of an Epsilon class glutathione transferase confers DDT resistance in the dengue vector, Aedes aegypti. Insect Biochem Mol Biol 2005, 35(8):861-871.
15. Marcombe S, Carron A, Darriet F, Etienne M, Agnew P, Tolosa M, Yp- Tcha MM, Lagneau C, Yébakima A, Corbel V: Reduced Efficacy of Pyrethroid Space Sprays for Dengue Control in an Area of Martinique with Pyrethroid Resistance. Am J Tropical Med Hyg 2009, 80(5):745-751.
16. Ranson H, Hemingway J (2005) Mosquito glutathione transferases. Methods in Enzymology 401: 226-241.
17. Raymond M, Prato G, Ratsira D (1997) Probit and Logit Analysis. Program version 2.0. Montpellier, France: Praxème R&D.
Rodriguez MM, Bisset J, De Fernandez DM, Lauzan L, Soca A: Detection of insecticide resistance in Aedes aegypti (Diptera: Culicidae) from Cuba and Venezuela. J Med Entomol 2001, 38(5):623-628.
18. Vontas JG, Small GJ, Hemingway J: Glutathione S-transferases as antioxidant defence agents confer pyrethroid resistance in Nilaparvata lugens. Biochem J 2001, 357(Pt 1):65-72.
19. Wirth MC, Georghiou GP: Selection and characterization of temephos resistance in a population of Aedes aegypti from Tortola. British Virgin Islands. J Am Mosq Control Assoc 1999, 15(3):315-320.
20. Yanola J, Somboon P, Walton C, Nachaiwieng W, Somwang P, Prapanthadara LA. High-throughput assays for detection of the F1534C mutation in the voltage-gated sodium channel gene in permethrin-resistant Aedes aegypti and the distribution of this mutation throughout Thailand. Trop Med Int Health 2011, 16:501-509.
21. WHO (2013) Test procedures for insecticide resistance monitoring in malaria vector mosquitoes. World Health Organization, Geneva.
22. WHO (2005) Guidelines for laboratory and field testing of mosquito larvicides. In: WHO/CDS/WHOPES/GCDPP/13, editor. Geneva, Switzerland: World Health Organization.
23. WHO (2006) Guidelines for testing mosquito adulticides for indoor residual spraying and treatment of mosquito nets. In: WHO/CDS/NTD/WHOPES/GCDPP/3, editor. Geneva, Switzerland: World Health Organization.
24. World Health Organization, 2004. Spinosad DT in Drinking-Water: Review of VectovacWG. Document WHO/CDS/WHOPES/2004.8. Geneva: World Health Organization.
25. World Health Organization, 2008. Diflubenzuron in Drinking-water: Use for Vector Control in Drinking-water Sources and Document WHO/HSE/AMR/08.03/6. Geneva: World Health Organization.
26. World Health Organization, 2010. Spinosad DT in Drinking-Water: Use for Vector Control in Drinking-water Sources and Containers. Document WHO/HSE/WSH/10.01/12. Geneva: World Health Organization.









